High quality lentiviral vector production
With over 17 years of know-how in lentiviral technology and a robust and proprietary bioproduction process, Flash Therapeutics has produced more than 9000 lentiviral vector batches for scientists and clinicians all over the world.
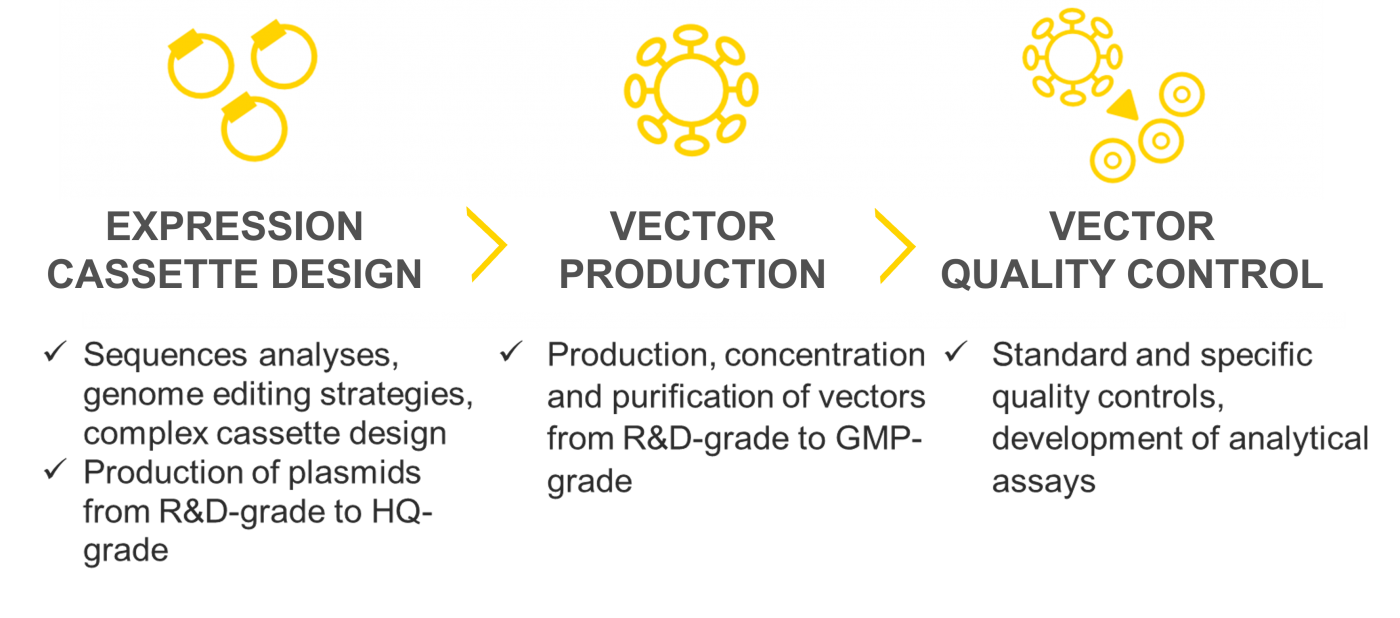
Flash Therapeutics can support you for the design of your project, then we take care of all steps to provide integrative lentiviral particules or LentiFlash® particles for RNA transfer.
Flash Therapeutics offers small, medium and large-scale productions of high-quality vectors from R&D grade to GMP grade.
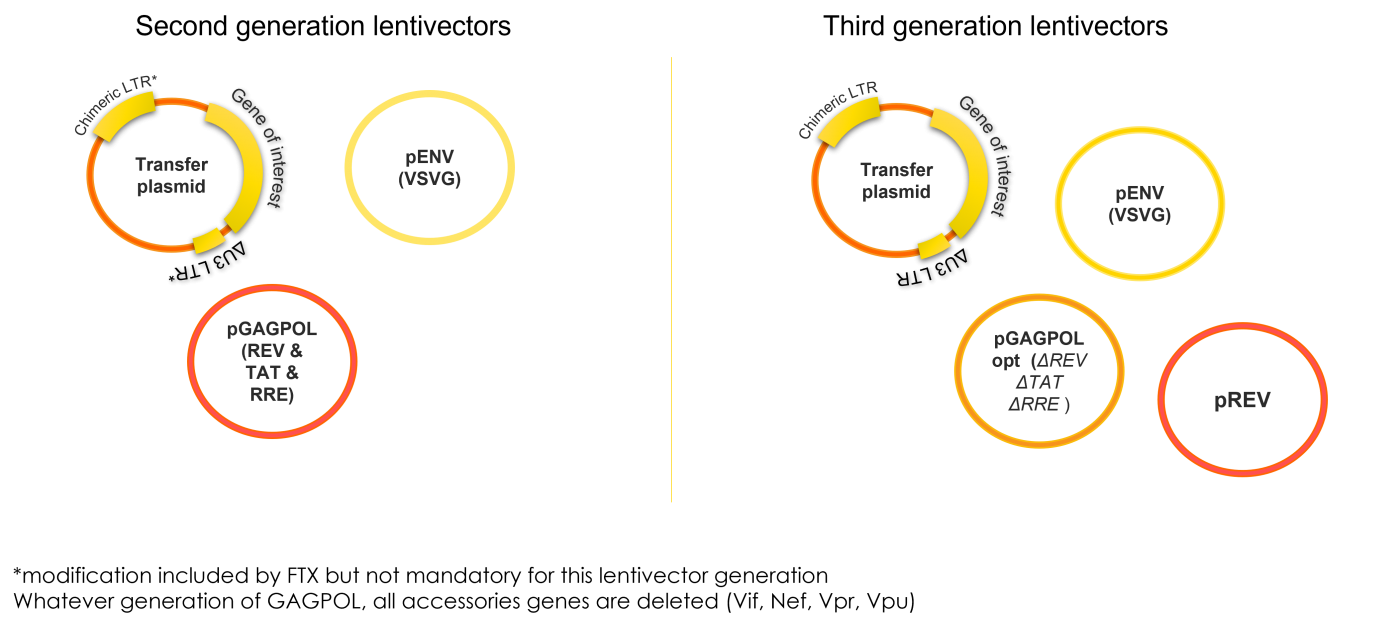
2G or 3G lentiviral vector depending on your needs
Three plasmids for second generation (2G) or four plasmids for third generation (3G) will be used to produce lentiviral particles. Helper Plasmids have been optimized (removal of unnecessary viral components, codon optimization…).
If needed, we can support you with the optimization of your expression construct as well.
These improvements significantly increase the safety profile of our lentiviral vectors, which can be used in category II laboratories in most of the EU and other countries. We can produce 3G vectors for research purposes also, to facilitate future clinical translation.
A robust lentiviral vectors production & purification
Investment in manufacturing started early in our development, as we consider that providing high quality products is a success factor for the development of cell and gene therapy but also at the discovery level, to ensure robustness of data obtained and their possible clinical translation.
Our lentiviral manufacturing platform provides the highest purity on the market.
A premium production process for premium vector quality
- Adherent cells (HEK293T)
- Serum and antibiotic free media, to limit contaminants
- CS or HS or Bioreactor, according to the final volume needed
- Several successive purification steps
- Single use technology
Flash Therapeutics production process allows removal of most of proteins and DNA impurities. This high purity level avoids phenotype changes or unwanted cellular responses following transduction.
Flash Therapeutics also developed a reliable and discriminating titration method based on the actual content of effective particles, quantified by qPCR in integrated Virus Genome units (IG). This ensures that we do not include in our titles the total number of particles including damaged or empty particles and guarantees robust and reproducible performances of vector batches.
We can also give a PP/IG ratio (on demand) which provides a good measure of the quality of the vector produced. This is part of our requirements for transparency and quality.
GMP manufacturing and purification of selected and optimized candidates.
As a CDMO, Flash Therapeutics has its cGMP compliant lentiviral vectors production platform for clinical applications, based on its know how on high quality vectors production.