2023-11-22Posters
by Alexandra ICHE

FlashRNA® is a non-integrative viral RNA delivery system allowing transient, efficient and safe gene expression which marks a significant milestone in the field of gene and cell therapy. Its non-toxic, non-immunogenic nature, high cellular uptake, and ability to transduce various RNAs showcase its value. This versatile technology, compliant with Good Manufacturing Practices (cGMPs), creates new therapeutic possibilities with enhanced safety features.
2023-03-21Posters
by Lucille Lamouroux

Discover the scientific poster presented at the Advanced Therapies 2023 congress in London, about our manufacturing process through a continuum from the R&D to the clinic.
2023-03-03Essentials
by Régis Gayon

Selecting the right CDMO to get lentiviral vectors according to the cGMPs is an important choice for a cell and gene therapy company, but very hard to handle. Choosing a CDMO with strong expertise and experience as a lentiviral vector manufacturer is crucial but not enough to be chosen as the best CDMO.
Here are the top five criteria to consider when making your choice.
2022-11-04Essentials
by Florine Samain

2022 has already been a record year for the approval of new gene therapies to treat rare diseases and cancers. More than 2,000 trials are ongoing at the end of June 2022, a slight decrease from the end of 2021. North America leads with about 40%* of active clinical trials, and Europe ranks third with 16%*.
Cell-based immuno-oncology (CAR T cells) represents 34%* of active clinical trials and gene therapies 18%*.
2020-10-12Essentials
by Nicolas Martin

Over the past decades, the manipulation of genes has led to the development of many methods to generate numerous genetically engineered mouse models for studying human diseases. One major technological leap was achieved by the site-specific recombination technology mediated by the Cre recombinase and its specific DNA “Lox” sites.
2020-03-30Essentials
by Christine Duthoit

At the end of 2018, for the first time in history, the two Chinese twins Lulu and Nana were born with their genome modified by the CRISPR / Cas9 system. This outraged the entire scientific community and ask us about the risks of therapies targeting Human genome. The Cas9 nuclease is often compared to DNA scissors, allowing a very precise cut into any gene. CRISPR are the hands that position these scissors at the desired site of the genome. This system promises to be able to repair any defective gene, thus appearing as the solution for many genetic diseases. It is a valuable tool as well for many therapeutic approaches using genome engineering.
2017-07-17Essentials
by Christine Duthoit
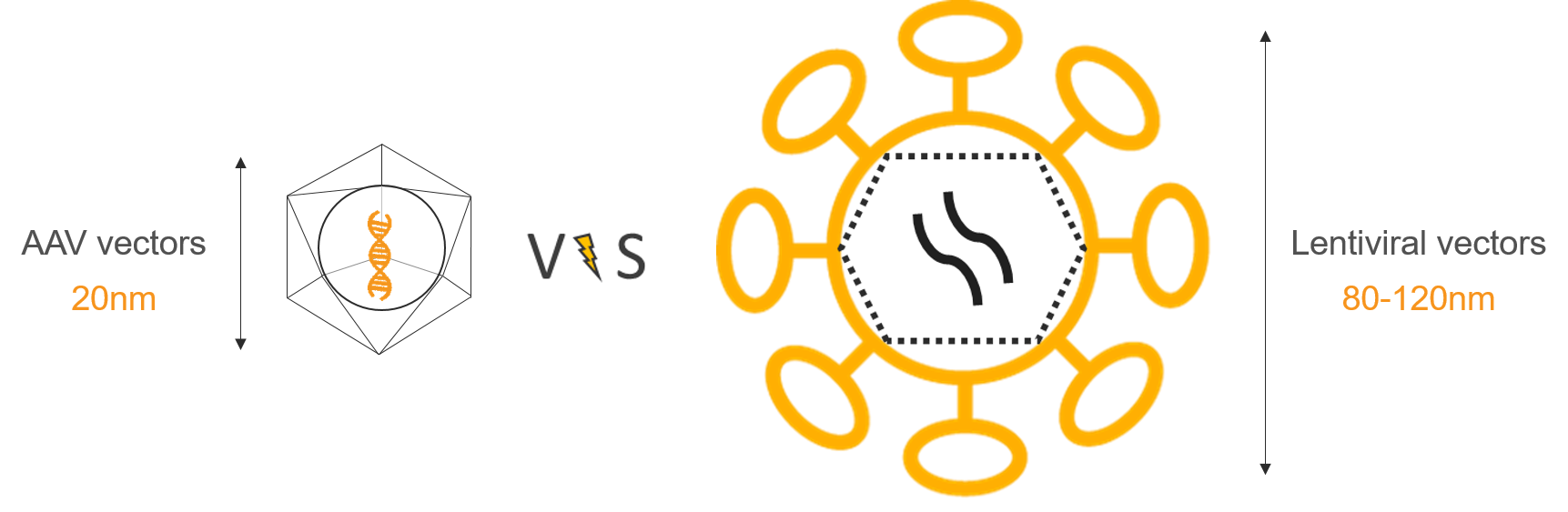
Let’s say you have one or several great gene candidates, you are done with all the in vitro studies and you have been asked to validate your results in vivo. Looking at the bibliography, you inevitably fall into thousands of data showing wonderful results using the Adenoviral Associated Vectors (AAVs). Now you should be asking yourself “what serotype do I need to choose for my project?” and there you go back to bibliography trying to find out a clear answer to this question… and you won’t.
2016-08-04Cell line generation
by Raphael Sevrain
- Electroporation,
- Lipid or chemical transfection,
- Viral vectors…
A wide range of tools is available for gene transfer experiments. Among these, the lentiviral vector technology has proven to be an effective tool with significant benefits. This is mainly due to its efficiency, its gentleness with delicate cells, the stability of expression and the shortened time lapse of experiments. Unfortunately, results are not always living up to expectations. To prevent failure, several parameters must be taken into account. We describe in this article 10 best practices to follow for a good transduction efficiency.
2016-06-27Essentials
by Pascale Bouillé
Delivery, delivery, delivery!
Dr Jennifer Doudna opened the last American Society of Gene and Cell Therapy Annual Meeting in Washington by highlighting the current challenges of gene editing :
• Delivery
• Controlling repair pathways
• Ethics
2016-05-09Cell line generation
by Pascale Bouillé

Lentiviral vectors insert the sequence of interest right into the host DNA thus allowing for a stable and long-term expression of the transgene. The gene will be copied every time the cell divides.










